TheCOVIDBlog.com
March 21, 2021

GUALEGUAYCHÚ, ENTRE RÍOS — An 85-year-old Argentinian woman is dead two days after receiving an experimental COVID-19 viral vector shot.
Mrs. Julieta Raquel Dumón received the shot on March 15. A photo of Mrs. Dumón, also called Julieta Dumón De Majul, was posted on Facebook by her son Alejandro Majul on that day.
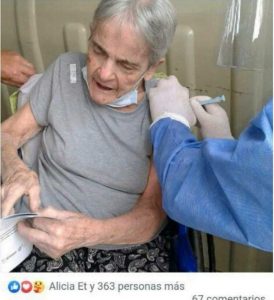
It is unclear which shot Mrs. Dumón received. But as of February 21, Argentina had 1.2 million doses of the Russian Sputnik V viral vector shots. It also had 580,000 doses of the Oxford-AstraZeneca shots. Argentina authorized the China-made Sinopharm inactivated virus shots on February 21. The latter, along with the Sinovac and Bharat Biotech (Covaxin) shots, are the closest things to “traditional” vaccines for COVID-19. However, all of them are experimental and still in clinical trials. Mrs. Dumón most likely received the Sputnik V shot, as it has been the most readily-available in Argentina since January.
Mrs. Dumón’s daughter, Julieta Majul, posted an update on Facebook two days later. Her mother passed away.
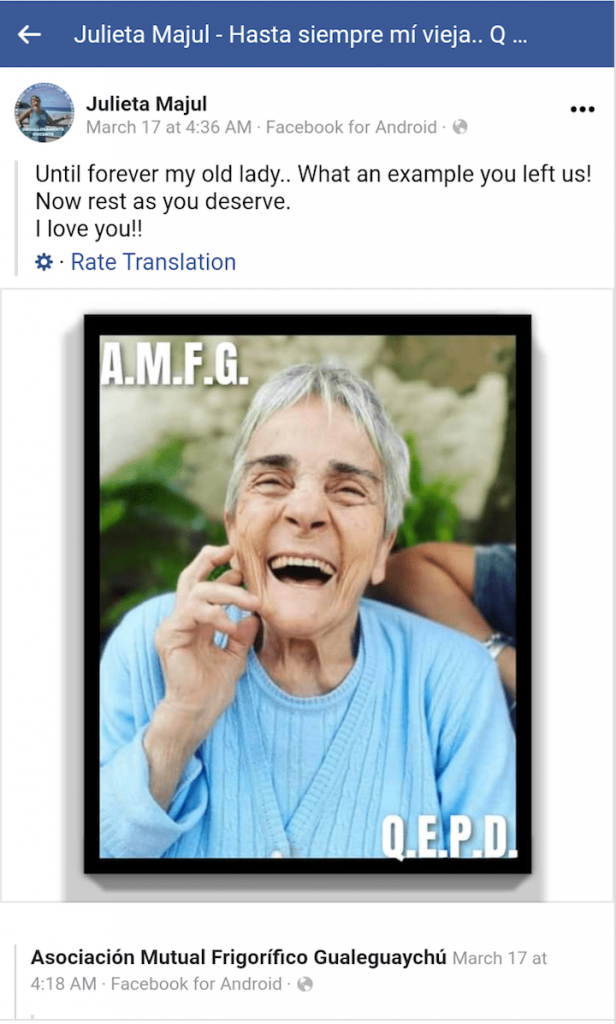
Her death was also announced on El Argentino.

Note that the “VDA” in her name is an abbreviation for viuda, meaning widow.
Stop inoculating the elderly
We have chronicled more than enough evidence showing that all these experimental shots are very dangerous for senior citizens. Parents and grandparents are stubborn and set in their ways. But it is your responsibility to protect your loved ones from these experimental shots.
COVID Legal USA is your partner in fight mandatory vaccines and other COVID mandates. Follow us on Telegram and Twitter. Read about white pine needles and order them here.
Fight back against censorship! We are once again processing credit card donations. CLICK HERE TO DONATE VIA CREDIT OR DEBIT CARD.


