TheCOVIDBlog.com
August 23, 2021 (document links updated March 1, 2023)

UPDATE September 1, 2021 – FDA Director of Vaccine Research and Review, Dr. Marion Gruber, and her top deputy, Dr. Philip, are stepping down from their positions in October and November, respectively. Gee, I wonder why? In other news, The Biden Administration is sidestepping the FDA, announcing it will unilaterally make booster shots available on September 20.
—ORIGINAL STORY—
SILVER SPRING, MARYLAND — The U.S. Food and Drug Administration (FDA) is an official subsidiary of Pfizer. The FDA granted full approval for the experimental Pfizer mRNA injections this morning. The agency also announced that the injections are being rebranded and called “Comirnaty” for marketing purposes.

Note that Pfizer has used the brand name Comirnaty for its mRNA injection in Europe since December 2020. The generic, non-proprietary name of the drug is tozinameran. But “Comirnary” branded injections are not available in the United States as of publishing. The World Health Organization (WHO) also listed Comirnaty on its list of emergency use injections on December 31, 2020. Thus the name isn’t new. It’s just never been disseminated much until now. It’s the same tactic AstraZeneca (“Vaxzevria”) uses to attempt hiding the deadly nature of its experimental viral vector DNA injections. AstraZeneca is called “Covishield” in India.
Here is part of the FDA statement from this morning regarding the Pfizer approval:
“The FDA’s approval of this vaccine is a milestone as we continue to battle the COVID-19 pandemic. While this and other vaccines have met the FDA’s rigorous, scientific standards for emergency use authorization, as the first FDA-approved COVID-19 vaccine, the public can be very confident that this vaccine meets the high standards for safety, effectiveness, and manufacturing quality the FDA requires of an approved product.”
What this all means
The FDA approved Pfizer’s Biologics License Application (BLA). A BLA is the procedural process for “full approval” via the FDA. Pfizer may now directly market the mRNA injections to the public instead of relying on the government and media to push its agendas.

The mRNA injections can also now be sold in interstate commerce instead of governments buying up all the shots. The approved BLA will also increase Pfizer mRNA demand as it’s unlikely any new COVID-19 injections will receive emergency use authorization (EUA) since one is now fully approved. More mandates are also likely now that Pfizer has an FDA-approved product,
Interestingly, the FDA granted Pfizer’s BLA but also extended its emergency use authorization (EUA) for “certain uses that are not included in the approved BLA.” That provision is specifically in reference to the EUA for children age 12 to 15. Further, Pfizer and the FDA conceded that “Comirnaty” and the EUA Pfizer injections are not the same product.

Pfizer also said in a statement that the Comirnaty-branded injections “will not be manufactured” and that Comirnaty products “are not orderable at this time.”
To be crystal clear, Comirnaty branded injections received the BLA (“full”) approval. The Pfizer-BioNTech branded injections are still under the EUA. They are “legally distinct” products, according to the FDA.
This news changes only two things. First, it provides vaxx zealots celebration material and new talking points for defending their holy sacrament. It’s also a sure sign that those who refuse to be poisoned must be ready and willing to protect their lives and well-being by any means necessary.
This “full approval” was essentially purchased by the federal government. Pfizer profits will exceed $33.5 billion in 2021 from the mRNA injections alone, mostly courtesy of the U.S. and European governments. The facts about mRNA injections, the FDA and the Centers for Disease Control (CDC), however, remain unchanged. Keep this article handy to share with your Fauci-worshiping friends and family.
mRNA is gene therapy
Let’s be clear – the Pfizer and Moderna mRNA injections are the same product. The Moderna June 30, 2020 Quarterly Report filed with the U.S. Securities and Exchanges Commission tells you exactly what the mRNA injections are on page 70:
Currently, mRNA is considered a gene therapy product by the FDA. Unlike certain gene therapies that irreversibly alter cell DNA and could act as a source of side effects, mRNA-based medicines are designed to not irreversibly change cell DNA; however, side effects observed in gene therapy could negatively impact the perception of mRNA medicines despite the differences in mechanism. In addition, because no product in which mRNA is the primary active ingredient has been approved, the regulatory pathway for approval is uncertain.
Despite their mitigation attempts with the foregoing word salad, mRNA injections are in fact gene therapy.
FDA official policy since 1984 prohibits casting doubts about “vaccine” safety
For this we look at the Federal Register, Vol. 49, No. 107, from June 1, 1984. Long story short, the FDA eliminated a federal testing provision (21 CFR § 630.11) to ensure oral polio vaccines remained a profitable endeavor. The agency said:
In tort litigation involving the Federal government and private parties, questions have been raised concerning whether some of the lots of vaccine used in the 1961 and 1962 clinical trials met the test standard for neurovirulence prescribed in §630.16(6)(1). The purpose of the neurovirulence tests, which is performed in monkeys, is to assure that the live virus used in the oral poliovirus vaccine is properly attenuated (non-virulent).
But wait, it gets better:
The FDA has reviewed the data and has concluded that, although there may be a question as to whether the results of all the neurovirulence tests met the standard in the regulations, there is no doubt that the oral poliovirus vaccine used in the clinical trials involving 195,000 subjects was acceptably low in neurovirulence.
21 CFR § 630.11 read (past tense) as follows:
To qualify for license, the antigenicity of the vaccine shall have been determined by clinical trials of adequate statistical design conducted in compliance with part 56 of this chapter, unless exempted under Sec. 56.104 or granted a waiver under Sec. 56.105, and with part 50 of this chapter. Such clinical trials shall be conducted with five lots of oral poliovirus vaccine that have been manufactured by the same methods. Type specific neutralizing antibody for each type of poliovirus in the vaccine shall be induced in 90 percent or more of susceptibles after a series of doses.
RELATED: CDC Foundation is not a government entity, has many conflicts of interest (March 23, 2021)
The FDA concluded its official declarations with the most infamous, totalitarian verbiage that is relevant in 2021:
However, although the availability of the vaccine may not be in immediate jeopardy, any possible doubts, whether or not well founded, about the safety of the vaccine cannot be allowed to exist in view of the need to assure that the vaccine will continue to be used to the maximum extent consistent with the nation’s public health objectives.
In other words, it is official FDA policy to lie about “vaccine” safety to ensure maximum sales. Here is a screenshot of the foregoing quote.

The following linked file is nearly 70 MB so it may take a couple minutes to download. But click here to read the full 1984 Federal Register yourself (it skips right to the relevant pages).
1976 Swine Flu vaccine halted after only three deaths
We’ve already written about this. But the 1979 60 Minutes broadcast talking about it is well worth the watch again in these surreal times.
RELATED: 60 Minutes: 1979 Swine Flu investigation has uncanny resemblances to COVID 2020-21 (February 26, 2021)
One item we left out of our original article is the 1976 New York Times piece. Nine U.S. states halted the use of the swine flu vaccines after just three people died that year. All of them had heart disease, but back then, that did not matter. The vaccine were considered unsafe and pulled off the market in Alaska, Illinois, Louisiana, Maine, New Mexico, Texas, Vermont, Virginia and Wisconsin. Allegheny County, Pennsylvania (Pittsburgh) also halted the injections. The 1960s polio vaccines killed 11 children and paralyzed 200 more. But remained on the market because polio was actually truly deadly, unlike COVID-19.
Nowadays Fauci and vaxx zealots proclaim that the Vaccine Adverse Event Reporting System (VAERS) is inaccurate. There have been 9,027 deaths reported in VAERS as a result of Pfizer injections alone as of August 13.
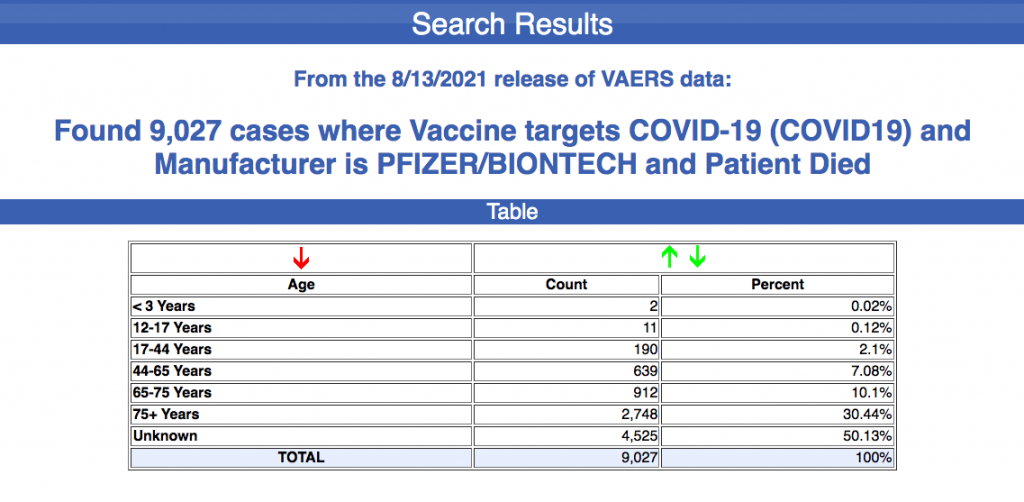
Let’s say, for the sake of argument, that only 10% of those reported deaths are accurate – a very generous error margin for the pharmaceutical industry at-large. That’s still 902 deaths, or 3,000% more than what triggered the cancellation of swine flu vaccines in nine states back in 1976. Times sure have changed.
Clinical trials are still ongoing
Don’t quote this blogger on the following statement. But this experimental Pfizer mRNA injection may be the first drug ever fully approved by the FDA with clinical trials still active. You may look at the full list of 32 Pfizer mRNA clinical trials that are either active or recruiting people right now. But here are a few highlights:
- The Safety of Administering a Second Dose of a COVID-19 mRNA Vaccine in Individuals Who Experienced a Systemic Allergic Reaction to an Initial Dose. Estimated Completion Date: December 30, 2022
- Patient Experiences With the COVID-19 Vaccination After Breast Cancer Treatment (LymphVAX). Estimate Completion Date: December 31, 2022
- Study to Describe the Safety, Tolerability, Immunogenicity, and Efficacy of RNA Vaccine Candidates Against COVID-19 in Healthy Individuals. Estimated Completion Date: May 2, 2023.
- Study to Evaluate the Safety, Tolerability, and Immunogenicity of an RNA Vaccine Candidate Against COVID-19 in Healthy Children <12 Years of Age. Estimated Completion Date: September 27, 2023.
You’d think this information would be vital and necessary for government regulators to fully approve a new drug that has killed hundreds of thousands, and perhaps, millions of people across the globe already. But not in this dystopian world.
We’ll be updating this article throughout the week.
COVID Legal USA is your partner in fight mandatory vaccines and other COVID mandates. Follow us on Telegram and Twitter. Read about white pine needles and order them here.



Rules simply don’t matter anymore.
“Appeal to authority” now on steroids.
“In other words, it is official FDA policy to lie about “vaccine” safety to ensure maximum sales.”
—————————————-
Welcome to Crony Crapitalism, courtesy of the Neoliberal / Neocon fraudster tag-team.
As you correctly note, the entire process of this (literally) manufactured virus has been one lie stacked upon another lie, and the power elite across media, the political and corporate realms have closed ranks to dutifully enable and guide the deception, as a for-profit enterprise.
I watched Chris Wallace / Fox news Sunday this weekend, who is supposedly the gold standard of fearless journalism, asking the “hard questions” of the power elite.
He interviewed the Surgeon General – Vivek Murthy – who is beyond any credibility due to his own conflicts of interest directly related to the mRNA vax’s. Murthy actually founded a private business dedicated to running trials for which Pfizer was a customer – meaning that he cannot under any circumstance act as an impartial or trusted public health overseer without disclosing all of his financial entanglements.
Murthy’s company has direct financial ties to the mRNA (and V.V.) vax makers, which you will only learn from doing your own personal research, because this has not been noted in a single major media report that I have come across. This is not merely an oversight – its deliberate deception on a scale that is ruthlessly maniacal.
Chris Wallace’s interview was a puff piece, as he covered everything that is NOT on the minds of average americans, but are central to the concerns of the power elite who want you injected, but dont have any interest in disclosure, or in holding those accountable who developed, funded and released this manufactured virus.
Wallace did not challenge Murthy on his financial profit from relations with the vax manufacturers – he never mentioned this issue.
In the end, ‘freedom of the press’ is totally meaningless – if not actually a ball and chain to the average citizen – if what you are left with is a corrupt cartelized entity that serves the interests of its ownership class as they pummel the average citizen into submission with their omissions, lies and deception.
Without knowing who is paying to control the mass media, and full ethical disclosure of these relationships, along with an expectation that those who have a conflict of interest recuse themselves, you end up in the society that we now have in which a corrupted criminal class of fraudsters unite to propagate their own interests over those who have been subjected to their domination.
we all most stand up and stop complying, Every time I hear about the protests, whether it’s parents fighting for mask free school attendance, or Nurses not being forced to take the jab, or any employees refusing to get it to keep their job, It makes me feel a little more hope. It’s going to take us standing our ground and not being afraid to lose our jobs. We need to plan ahead now. We need to be ready to fight. We need to be ready to take care of ourselves. and in turn be there for those of like mind. We need to build communities NOW. We need to Leave their evil system of control behind.
I’ve made some preliminary plans with some relatives who have crop land and beef cattle and are skilled at hunting wild game. I suggest everyone start thinking this way.
We’ve been thinking this way, but don’t know anyone. Happy you have found some options, though. We’ve been stocking up on canned goods (e.g. sardines) and other items, but we should probably buy a Berkey filter or something, since water will be the limiting resource.
You are not obligated to comply with any mandates. They are not legal, not laws. They’d also have to prove covid 19 exists in the first place. They can’t. Every bit of this is fraud. Demand proof. Fight like your life depends on it, because it does.
Forced vaccination is illegal, even asking about it violates HIPPA, some claim it violates the Nuremberg Code. Medical records, and treatments are supposed to be private, being forced to share that information, with an employer, as a term of employment violates a whole plethora of state, and local laws.
Currently many employers are having a difficult time recruiting new employees, thanks to the government cheques being handed out everywhere, but even if that wasn’t the case training a new employee for any job costs money. Doctors, nurses, and skilled tradesmen are always in short supply, they can always find a job elsewhere, and good luck finding replacements. Is it really worth sacking one, leaving a large hole in an organization, just to make some stupid point of wokeness, over vaccines that are increasingly proven not to be that effective?
They will kill us all. This is depopulation!
“Official data shows AT LEAST 32,000 people have died after having the Covid-19 Vaccine in Brazil, and these are just the deaths that have been labelled Covid-19”.
That Deagel 2025 population forecast is looking more and more plausible every day.
Now the idiots who were screaming on rooftops, to sound smart : “I am not getting vaxxed because it hasn’t been approved by the FDA”, can go and get their “approved” vax. I will be cheering for them.
Minus 13 ??!!. That’s too much. There should be a “Max Hate Allowance” in the comment section.
Call it a hunch, but I am reasonably sure that many people were using that as an explanation that even the sheep could not argue with. I can certainly relate to that thought, but does not mean it was their only reason.
An article on this site gave what I think is the strongest reason why these shots cannot be forced on anyone that does not want them.
https://thecovidblog.com/2021/04/27/supreme-court-pfizer-moderna-et-al-may-own-your-genes-once-youre-injected-with-their-lab-created-mrna-dna/
If I prefer to remain free from the dangers of ADE and decide the best way to do that is to avoid all injections, then as free person, that should be my right. To force me to follow some completely unproven course from a corporation makes me a slave to their technology. As far as I know, the 13th Amendment has not been formally revoked.
Who’s running to get the “approved” vax?
Wasn’t the FDA the same body that approved thalidomide for morning sickness, and AZT for treating HIV? Are they not the same ones, or was that the CDC, that disapproved of doctors using Hydroxychloroquine and Zinc, and Invermectin as preventatives, or for treating the CCP virus, in spite of there being no deaths from using either correctly?
I recall last year some Yahoo “medical reporter” interviewing some gloating Ron Burgandy-looking kook from the FDA and Duke Univ after the EUA for HCQ was removed, so I assume it was the FDA that did it.
On the bright side, I have heard of a DR in Cali that prescribed the neighbors of a friend the following for a c19 diagnosis: HCQ, NAC and ivermectin, along with C, D and Zinc and an antibiotic. So it seems even in the hellish smoking CA landscape there is a ray of hope. My friend and the neighbors are all unvaxxed.
I’m sure no money changed hands during all this…
I think it does make a huge difference. The vaccines were licensed for Emergency Use only which can only be granted (unless I’m quite wrong) if there is no alternative treatment. That’s why there is an ongoing obvious conspiracy against Ivermectin, for example.
Dozens of peer reviewed studies testify to the safety and efficacy of Ivermectin and the fact that it would have prevented UP TO 83% of deaths from Covid-19. So I calculate that by banning it they caused up to 3,300,000 extra deaths.
A veritable holocaust to secure mega profits. When will mass media publish this !
Now that it’s ‘Fully Approved’ they don’t need to renew their Emergency Use permit, at least for Pfizer, though they might still need it for the 3rd and 4th booster shots.
Does this mean that Pfizer’s lawsuit protection is over ? Nobody can say that long term safety studies can be done in less than a year. So, this is a rubber stamp, not regulatory oversight. Pfizer and the FDA should be sued. Otherwise, let me sell that Brooklyn Bridge. Its mine, you know. Clear title and everything.
It’s all bullshit…..the vax that is approved is the one with the name it sounds like community….but it’s not available in the us. The Pfizer one that everyone has gotten is still under eau and there is liability protection on that one but not on the approved one…..it’s all smoke and mirrors.
Now, the big pharma looting operation is totally blessed by the government sycophants. They have just approved a very dangerous product that has killed hundreds if not thousands of people and harmed many many more. Is this about culling the population? Because it certainly isn’t about health.
Why have so many doctors forgotten their Hippocratic Oath and especially the first tenet of it, ‘do no harm?’ I have never trusted the jackals in Big Pharma and now I don’t trust the Medical Community.
Simply put, money talks, BS walks.
“The love of money is the root of all evil”.
– 1 Timothy 6:10.
You can stick your fake vaccine up your arse!!!!
It’s not a vaccine, it’s the sloppy administration of a poorly tested toxic drug called a spike-protein.
It’s functionally equivalent to:
1. Manufacture the spike-protein drug. We know how to do that. Purify it. Add some fillers to aid injection.
2. Randomly inject different amounts into individual patients, keeping no record of how much drug each patient got.
3. Do no active monitoring of positive or negative effects on individuals, only studying statistical preventative effects with regards to COVID.
4. No placebo control group.
The above scenario is equivalent to the injection of mRNA as a vaccine. The exact same drug is produced with no control over dose or the duration of its production bt the body. No useful measures of toxicity of the drug.
If any doctor tried to inject the spike protein into their patients they would lose their licence to practice medicine. If the consequences for any patient were severe, the doctor and probably their clinic could be sued.
But, because it’s called a vaccine, there are none of the standard scientific requirements of testing a new toxic drug.
Wow, well put Tracy. Even so, I suspect what is inside (besides the mechanisms that make cells produce the s-antigens) and what those substances will ultimately do, are far more sinister than we know.
Please correct this!!! They did not grant “full approval”, per Mike Adams!
Please read the article. Per The COVID Blog.
BLA, EUA at the same time…So, does this continue to shield them from liabilities? Does specific labeling shield them?
Polio was caused by DDT and the vaccines, THERE IS NO POLIO VIRUS! No vaccine in history has ever worked because there is no such thing as viruses that make you sick. All vaccines are poison. EVERY vaccine kills and maims, NONE of them are anything but poison. Pasteur was a fraud, and all of his experiments failed and killed people. The establishment took his failed research and lied about it to make money and to make people sick, sound familiar? If you care at all about the truth, then I implore you to follow the work of Dr Kaufman and Dr Cowan and start looking at the ACTUAL science behind the germ theory that has been soundly proven to be completely false. (And this means the current narrative is a TOTAL lie too, there is no virus) It’s an inconvenient truth that everything you’ve believed your whole life about being sick is a lie, but you’ve come far enough to realize you’re being lied to about these injections, don’t let cognitive dissonance keep you from the whole truth.
Comirnaty? : sounds like ‘Comorbidity’ – get yours free today !
Hi, thanks for getting the US truth out. There are many people that cannot get this injection. I’ve never heard of being sick or C + and bring told to go home for 2 weeks until you can’t breathe. Time is precious if you are sick. If the gvmnt is that concerned for our health, why have they not distributed preventative and early treatment kits to “we the people” ? Why is their only option the Holy Grail unapproved vaccine? DJT suggested HCQ when this started. He was chastized. US Doctors hands are tied to rx treatment. With IVM and combos this may have been eradicated long ago. To get truth you need to watch AU news? This is criminal. They apparently do not propagate the vaers dbs so we need to look at EUropes dbs. I’m appauled at this. I pray we will get preventative treatment/early treatment kits now in the US. People are dying and being crippled for life. I’d say, that’s not ethical nor economical. Thanks for putting data out there. We need all the logic and expertise of combined doctors dealing with this situation. When did the CDC and FDA become the 3rd political party in the US?
Don’t risk your life by taking the “jab” to save your job b/c in the end you will either die, or get very sick from the “jab”. Stand up for your God given rights for Body Autonomy.